Risk Management in Clinical Trials
This book is a ‘must read’ for anyone who needs to know about the general principles of risk management in clinical trials.
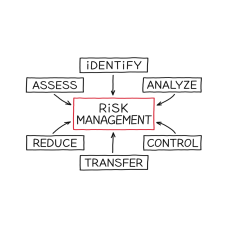
We utilize relevant regulatory guidance as well as the principles of ICH GCP to help you understand the risk management process and how this is done.
Content includes:
• the principles of risk-based management of clinical trials
• who should be involved in a risk assessment
• the identification of ‘Critical to Quality’ factors
• the steps involved in a risk-based approach to clinical trials
• setting quality tolerance limits
• risks at the system and trial levels
• risk-based monitoring
• case studies.
e-book Risk Management in Clinical Trials
- Product Code: e-book Risk Management in Clinical Trials
- Availability: In Stock
-
£15.00
Related Products
Risk Management in Clinical Trials
Risk Management in Clinical Trials In this 60-min course, we look at the general principles relat..
£100.00